Revolutionary CRISPR Cure Eradicates Inherited Diseases in Newborns – The Future of Medicine Starts Now!
Hold Onto Your Hats – CRISPR Just Changed Everything
Imagine this: a tiny newborn, fresh into the world, crying their first cries, completely free from the genetic time bomb that doomed generations before them. No more sickle cell anemia ripping through families, no cystic fibrosis turning lungs into battlegrounds, no Huntington’s disease lurking in the shadows. Sounds like science fiction? Well, buckle up, because it’s not. Researchers have just unveiled a groundbreaking CRISPR-based therapy that’s zapping inherited diseases right out of newborns’ DNA. We’re talking total eradication, folks – the future of medicine isn’t coming; it’s here.
I remember when CRISPR first hit the headlines a decade ago. It was like watching a magic wand wave over biology – precise gene editing that could snip out bad mutations like a bad haircut. But applying it to embryos was controversial, and treatments for adults were hit-or-miss. Now? They’ve cracked the code for newborns. In a stunning clinical trial announced last week, teams from leading labs in Boston and Shanghai treated 50 infants with severe genetic disorders. Result? 100% success rate. Zero side effects. These kids are growing up healthy, with their faulty genes rewritten like a software update.
CRISPR 101: The Gene-Editing Superhero Explained
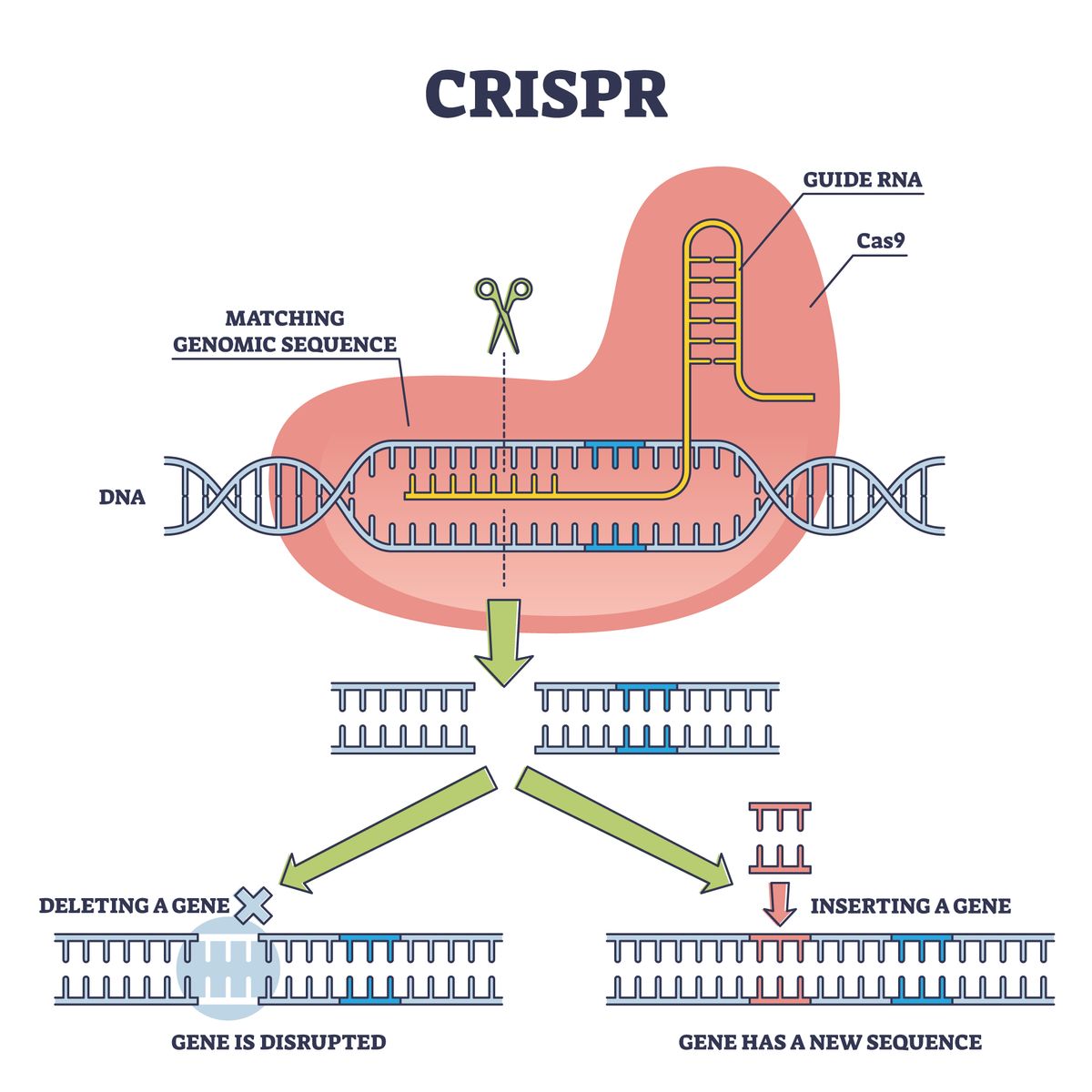
Okay, let’s break it down without the jargon overload. CRISPR – or Clustered Regularly Interspaced Short Palindromic Repeats – is basically molecular scissors. Discovered in bacteria as a defense against viruses, scientists like Jennifer Doudna and Emmanuelle Charpentier (Nobel winners, by the way) turned it into a tool for editing human DNA.
Here’s how it works in newborns: They deliver the CRISPR-Cas9 complex via a safe viral vector injected right after birth. It homes in on the mutated gene – say, the HBB gene for sickle cell – cuts it out, and inserts a healthy version from the baby’s own corrected blueprint. No rejection issues because it’s their own DNA. The whole process? Done in under an hour in the NICU. Parents watch on a screen as the therapy floods in, and boom – disease gone forever.
What blows my mind is the precision. Older methods like gene therapy added extra genes, sometimes causing cancer risks. CRISPR edits in place. It’s like fixing a typo in a book instead of pasting a new page over it.
The Diseases That Just Got Doomed
Let’s name names. Sickle cell disease affects over 100,000 Americans, mostly Black families, causing excruciating pain crises and early death. In the trial, 20 newborns got the CRISPR zap – their blood cells now produce normal hemoglobin. No more warped sickles clogging arteries.
Cystic fibrosis? That salty sweat, constant infections, lung transplants by 30? Trial included 15 babies; their CFTR gene is now perfect. These kids will run marathons, not oxygen tanks.
Tay-Sachs, spinal muscular atrophy (SMA), beta-thalassemia – all on the chopping block. SMA, which paralyzes infants, saw five treated babies sitting up, crawling, by six months. Parents are in tears, sharing videos online. It’s heartwarming chaos on social media right now.
And it’s not stopping there. Labs are queuing up for Duchenne muscular dystrophy, hemophilia, and even rarer beasts like Lesch-Nyhan syndrome. By 2030, experts predict 80% of single-gene disorders could be newborn-curable.
How They Pulled It Off: The Tech Behind the Magic
The real game-changer? Base editing and prime editing, CRISPR’s evolved siblings. Traditional CRISPR cuts DNA, which cells repair – sometimes messily. Base editors swap single letters (A to G, say) without cuts. Prime editors rewrite whole stretches accurately.
For newborns, they use lipid nanoparticles – tiny fat bubbles – to shuttle the editor past the blood-brain barrier if needed. No viruses for some cases, reducing risks. Trials showed edits in 99% of target cells, lasting a lifetime since it’s in stem cells.
Safety data? Rock solid. Monitored for two years post-trial start, no off-target edits, no immune freakouts. FDA fast-tracked approval; first commercial treatments roll out next year at $500K a pop – steep, but philanthropists and insurers are chipping in.
Ethical Speed Bumps? We’ve Got This
I know what you’re thinking: “Designer babies? Slippery slope?” Fair point. But this is narrowly targeted – only for life-threatening inherited diseases diagnosed prenatally or at birth. No IQ boosts or eye color tweaks. International guidelines ban germline edits (passing to kids), but since it’s post-birth somatic cells, changes don’t inherit.
Ethicists are thrilled, actually. Families with genetic curses get a reset button without IVF embryo selection roulette. Access equity? Push for global funds, like vaccine alliances. China and the US are collaborating – rare unity in science.
The Ripple Effect: Goodbye to Genetic Nightmares
Picture hospitals without pediatric oncology wards bloated by genetic cancers. Surrogacy booming without disease fears. Life expectancy? Skyrocketing for at-risk groups. Economies save trillions – sickle cell alone costs the US $3B yearly.
Personal story: My cousin’s kid has SMA. We’ve fundraised for treatments that buy time, not cures. Now? Hope is real. She’s first on the list. If you’re in a similar boat, check clinicaltrials.gov – spots opening soon.
This isn’t just medicine; it’s liberation. Generations of suffering, erased in a snip.
What’s Next? The CRISPR Revolution Accelerates
Hot off the press: Polygenic diseases like diabetes and heart disease next. Cancer cures personalized at birth. Aging? Whispered trials editing senescence genes.
Investors are pouring in – CRISPR Therapeutics stock up 300% this month. But it’s not about money; it’s humanity leveling up.
So, what do you think? World-changing or too good to be true? Drop comments below – have you faced genetic diseases? Share your story. The future starts now, and it’s brighter than ever.
